40 bohr diagram oxygen
The Bohr effect is a phenomenon first described in 1904 by the Danish physiologist Christian Bohr. Hemoglobin's oxygen binding affinity (see oxygen-haemoglobin dissociation curve) is inversely related both to acidity and to the concentration of carbon dioxide. That is, the Bohr effect refers to the shift in the oxygen dissociation curve caused by changes in the concentration of carbon ... draw a Bohr-Rutherford diagram for oxygen. draw a Bohr-Rutherford diagram for fluorine. a Bohr-Rutherford diagram is used to show the numbers and locations of protons, neutrons, and electrons in an atom. step 1. In atomic physics, the RutherfordBohr model or Bohr model or Bohr diagram, presented by Niels Bohr and Ernest Rutherford in , a system ...
Bohr Diagram Oxygen ... 960x720 0 0. Like JPG. Bohr Diagram For Oxy... 1466x1800 0 0. Like JPG. Bohr Model Oxygen Sc... 1024x768 0 0. Like JPG. Bohr Model Descripti... 600x400 0 0. Like JPG. Diagram Example Of A... 474x613 0 0. Like JPG. Diagram Of The Eye Q... 800x600 0 0. Like JPG. Diagram Of The Heart... 638x479 0 0. Like JPG. Do And Answer ...
Bohr diagram oxygen
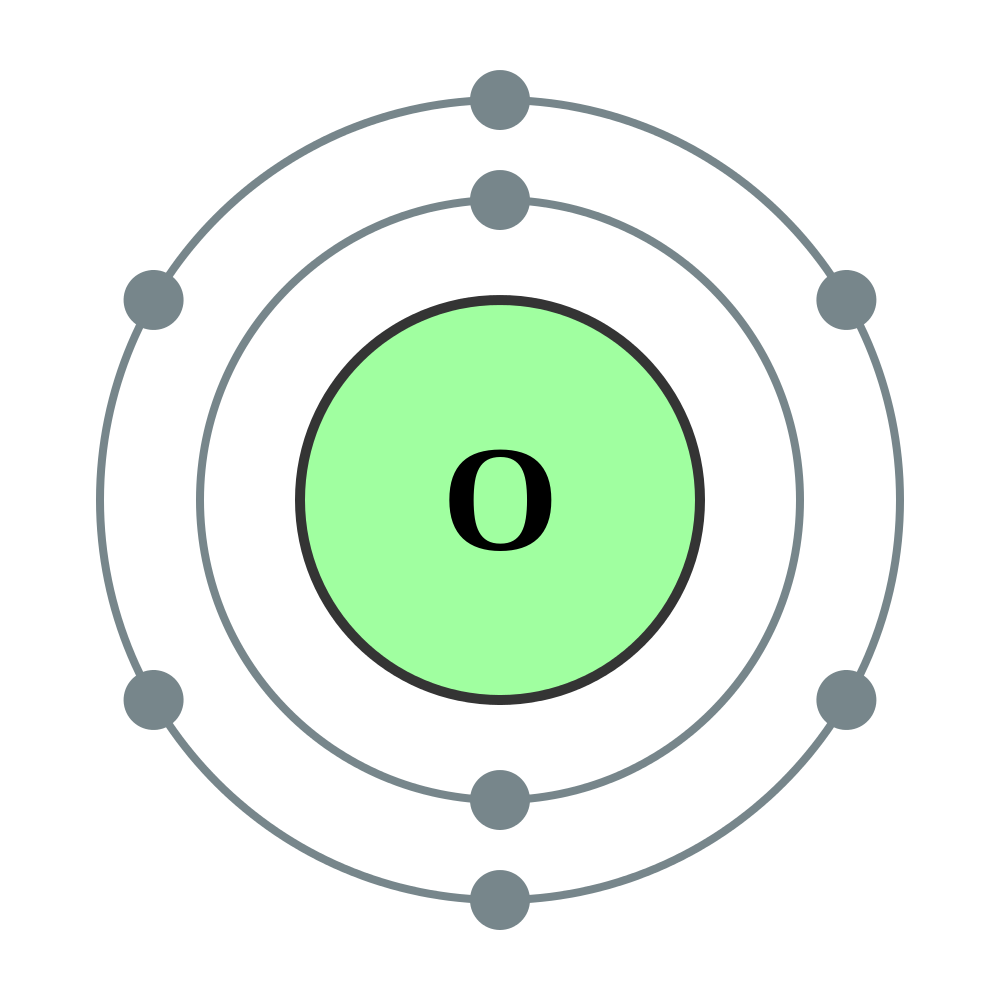
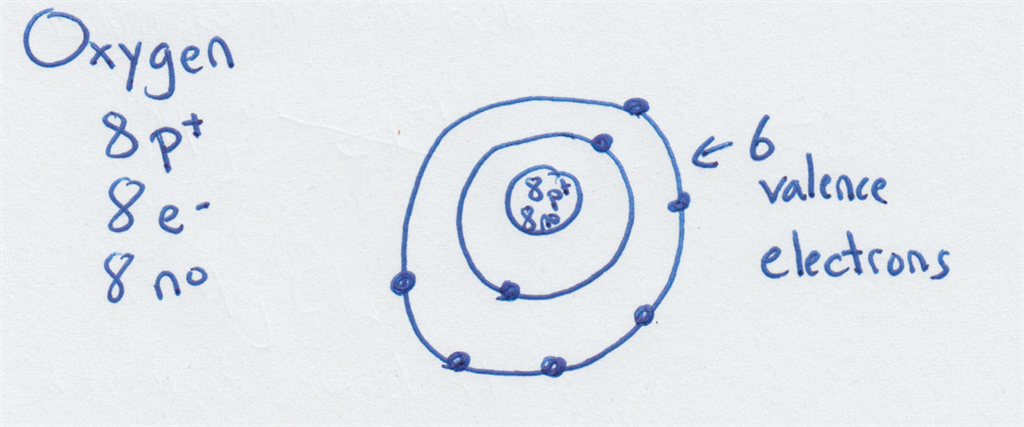
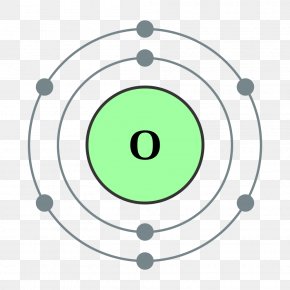
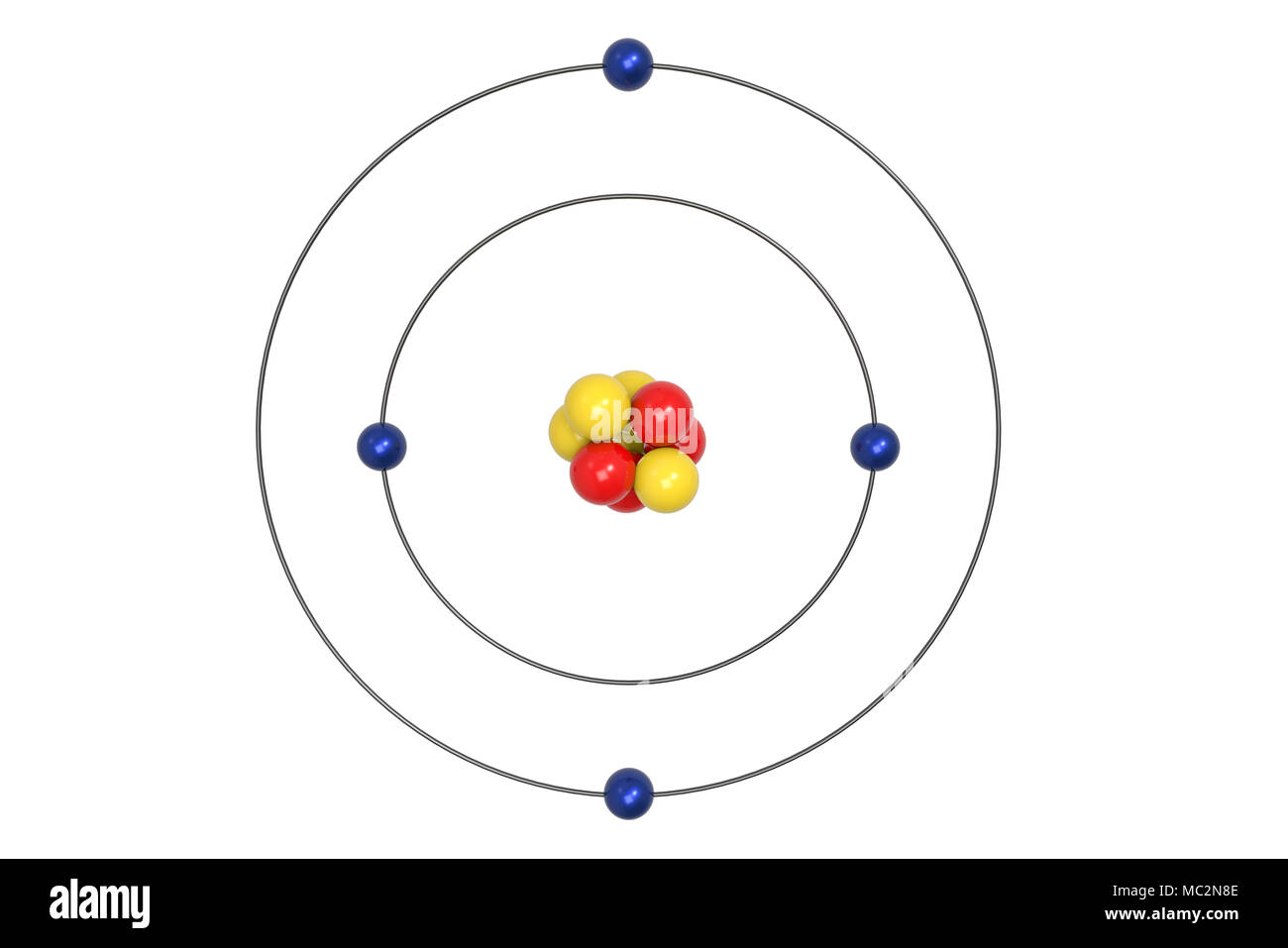
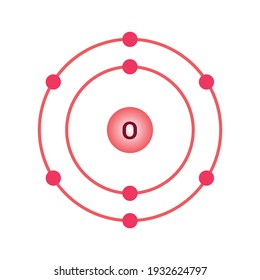
The bohr Rutherford diagram for oxygen has 8 protons and 8 neutrons. There are 2 electrons on the first orbital and six on the second. The bohr Rutherford diagram for oxygen h as 8 protons and 8 neutrons. Identification d'éléments à partir du diagramme de Bohr-Rutherford. Bohr - Rutherford Diagrams study guide by Mr_Vasiliou includes 26 ... Name: Oxygen Symbol: O Atomic Number: 8 Atomic Mass: 15.9994 amu Melting Point:-218.4 °C (54.750008 K, -361.12 °F) Boiling Point:-183.0 °C (90.15 K, -297.4 °F) Number of Protons/Electrons: 8 Number of Neutrons: 8 Classification: Non-metal Crystal Structure: Cubic Density @ 293 K: 1.429 g/cm 3 Color: colorless Atomic Structure The Bohr model for oxygen shows eight protons and neutrons in the nucleus of the atom, with eight electrons orbiting the nucleus in two energy levels....
Bohr diagram oxygen. Photo "Oxygen Atom Bohr model with proton, neutron and electron. draw a Bohr-Rutherford diagram for oxygen. Bohr developed the Bohr model of the atom, in which he proposed that energy levels of electrons are discrete and that the electrons revolve in stable orbits around the atomic nucleus but can jump from one energy level (or orbit) to another. Bohr Diagrams 1) Check your work. 2) You should have 6 total electrons for Carbon. 3) Only two electrons can fit in the 1st shell. 4) The 2nd shell can hold up to 8 electrons. 5) The 3rd shell can hold 18, but the elements in the first few periods only use 8 electrons. 6p 6n. Bohr Diagrams Try the following elements one at a time: a) H b) He answer choices. The bohr model diagram represents all the subatomic particles, while the lewis dot diagram only shows the symbol and the valence electrons. The lewis dot shows all the electrons and the bohr model only shows the electrons in the last shell. The lewis dot shows the number of neutrons and the bohr model only shows the electrons. Bohr found that an electron located away from the nucleus has more energy, and electrons close to the nucleus have less energy. Postulates of Bohr's Model of an Atom In an atom, electrons (negatively charged) revolve around the positively charged nucleus in a definite circular path called orbits or shells.
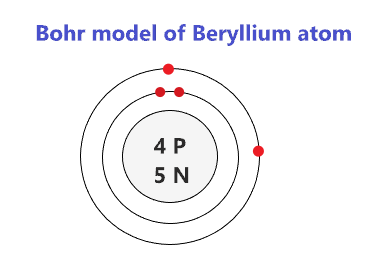
A Bohr diagram is a simplified visual representation of an atom that was developed by Danish physicist Niels Bohr in 1913. The diagram depicts the atom as a positively charged nucleus surrounded by electrons that travel in circular orbits about the nucleus in discrete energy levels. Oxygen Fluorine Nitrogen Silicon . Lewis Dot Diagram Worksheet Use the Bohr models to determine the number of valance electrons. Once you have found the number of valance electrons, place them around the elements symbol. Element Atomic # Atomic Mass Protons Neutrons ... An example of a Bohr-Rutherford Diagram for oxygen is shown in Figure #2: Remember: Protons (p+) and neutrons (n0) are shown in the nucleus (centre). 07/03/2021 · Bohr model of Helium (He) 2: 3: Bohr model of Lithium (Li) 2, 1: 4: Bohr model of Beryllium (Be) 2, 2: 5: Bohr model of Boron (B) 2, 3: 6: Bohr model of Carbon (C) 2, 4: 7: Bohr model of Nitrogen (N) 2, 5: 8: Bohr model of Oxygen (O) 2, 6: 9: Bohr model of Fluorine (F) 2, 7: 10: Bohr model of Neon (Ne) 2, 8: 11: Bohr model of Sodium (Na) 2, 8 ...
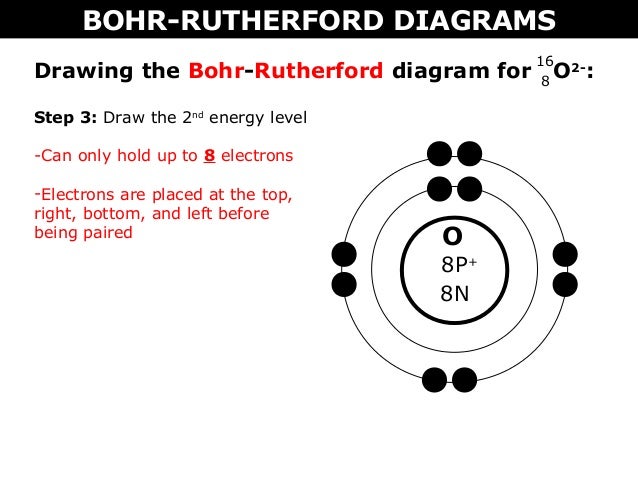
How to draw the Bohr-Rutherford Diagram for Oxygen. 2 electrons can go in the first shell, 8 in the second, 8 in the third, and so on... A Bohr diagram is a simplified visual representation of an atom that was developed by Danish physicist Niels Bohr in 1913. The diagram depicts the atom as a positively charged nucleus surrounded by electrons that travel in circular orbits about the nucleus in discrete energy levels. Additionally, what is the proton of oxygen? 8. Bohr Diagrams • A Bohr diagram is a diagram that shows how many _____ are in each shell surrounding the nucleus. • Named in honour of _____, a Danish physicist who developed several models for showing the arrangement of electrons in atoms. • There are three main background questions to explore before we start drawing Bohr diagrams. since air is a mixture of oxygen and nitrogen gases, both elements react with the magnesium metal. Graphic in a new window Magnesium Oxide: Determine the formula of a compound formed by the reaction of magnesium and oxygen.
Aug 15, 2020 — Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are ...
We collected 40+ Bohr Model Drawing Oxygen paintings in our online museum of paintings - PaintingValley.com. ADVERTISEMENT. LIMITED OFFER: Get 10 free Shutterstock images - PICK10FREE. oxygen. model. bohr. diagram. atomic. rutherford.
Bohr Model Practice 1. Carbon , 4. Chlorine 7. Aluminum 10. N'ì NAME BLOCK 3. Oxygen 6. Neon 9. Helium 12. Beryllium 2. Hydrogen 009 5. Sodium
20/05/2021 · Oxygen Delivery at Tissues. As shown on the diagram above, the percentage of oxygen bound to haemoglobin is related to the partial pressure of oxygen (pO 2) at a given site. When oxyhaemoglobin reaches a tissue that has a low pO 2 (e.g. skeletal muscle), it will dissociate into oxygen and haemoglobin, resulting in an increase in local pO 2. ...
*Iron does not follow the rules in determining the Lewis diagram. This is a special case and unless you are an expert, you would not be expected to figure this out by yourself. Write the Lewis symbols for each atom. See graphic on the left.
History and mathematical formalisms Christian Bohr and the concept of cooperative binding. In 1904, Christian Bohr studied hemoglobin binding to oxygen under different conditions. When plotting hemoglobin saturation with oxygen as a function of the partial pressure of oxygen, he obtained a sigmoidal (or "S-shaped") curve. This indicates that the more oxygen is bound to hemoglobin, the …
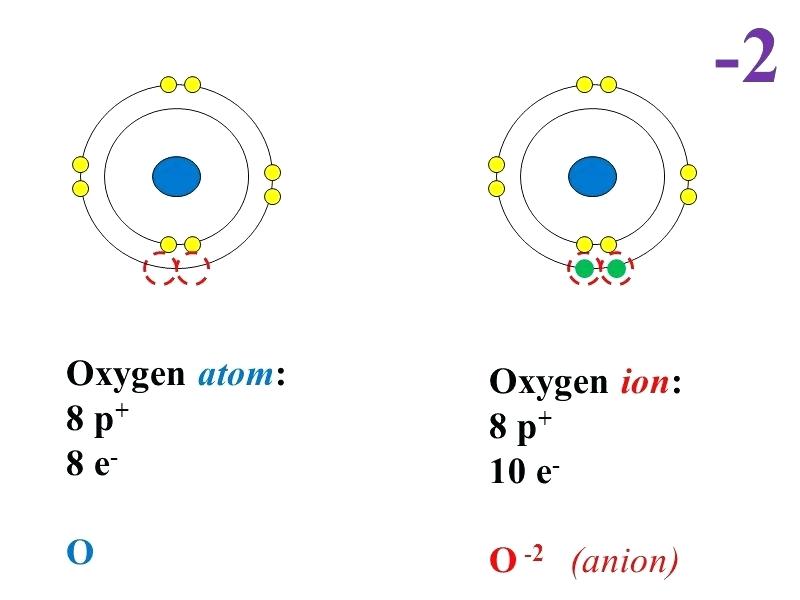
1 answerBohr diagram for Oxygen atom and Oxygen ion. An oxygen atom has 2 orbits, in the first orbit, there are 2 electrons moving in opposite directions...
Bohr Model of Hydrogen. The simplest example of the Bohr Model is for the hydrogen atom (Z = 1) or for a hydrogen-like ion (Z > 1), in which a negatively charged electron orbits a small positively charged nucleus. Electromagnetic energy will be absorbed or emitted if an electron moves from one orbit to another.
Bohr Model Drawing Of Oxygen At Paintingvalley Com Explore Collection Of Bohr Model Drawing Of Oxygen
07/06/2016 · Each gram of haemoglobin can combine with 1.34 mL of oxygen. At low partial pressures of oxygen (e.g. within tissues that are deprived of oxygen), oxyhaemoglobin releases the oxygen to form haemoglobin. The oxygen dissociation curve has a sigmoid shape because of the co-operative binding of oxygen to the 4 polypeptide chains.
15. Calculate the missing information and then draw the Bohr Diagram and Lewis Structure for each element. Mg Atomic # = 17 # of N = 16. Answer the questions below based on the elements in question #15. (1) Which elements had a filled outermost shell? He & Ne (2) Which element would be most likely to lose electrons in a chemical bond?
Oxygen electron configuration is 1s 2 2s 2 2p 4.The period of oxygen is 2 and it is a p-block element. This article gives an idea about the electron configuration of oxygen(O) and orbital diagram, period and groups, valency and valence electrons of oxygen, bond formation, compound formation, application of different principles. The eighth element in the periodic table is oxygen.
He & Ne Why? They have full outer shells. Bohr Diagram. Shows all electrons. Lewis Structure. Shows valence electrons.2 pages
Bohr Diagram Of Oxygen. how to draw bohr rutherford diagrams oxygen how to draw the bohr rutherford diagram for oxygen 2 electrons can go in the first shell 8 in the second 8 in the third and so on diagram oxygen bohr model diagram template information title oxygen bohr model diagram categories diagram ♦ publised sunday january 29th 2017 09 00 03 am
•When elements form compounds, changes occur in the arrangement of electrons in the outer orbit. • Electrons are gained or lost so that element can have a stable electron arrangement of the closest noble gas. • Atoms prefer a completely filled outer shell with electrons • In order for a compound to be stable, it must have a completely filled ...
24/09/2019 · Bohr Diagram: The First Element. In order to make a Bohr diagram, you need to know the number of protons, neutrons, and electrons the element has. In this section, we’ll show a sample Bohr diagram for hydrogen. H —Hydrogen. 1 proton. 1 electron. 0 neutrons

Oxygen Atom Bohr Model Rotate On White Background 3d Rendering Stock Video Download Video Clip Now Istock
Feb 15, 2016 - How to draw the Bohr-Rutherford Diagram for Oxygen. 2 electrons can go in the first shell, 8 in the second, 8 in the third, and so on...
Oxygen - Atomic Diagram Carbon Atom Bohr model with proton, neutron and electron. 3d illustration Bohr model of Nitrogen Atom with proton, neutron and electron.
Oxygen Atom With Proton Neutron And Electron 3d Bohr Model Illustration Stock Illustration Illustration Of Chemistry Neutron 105289725
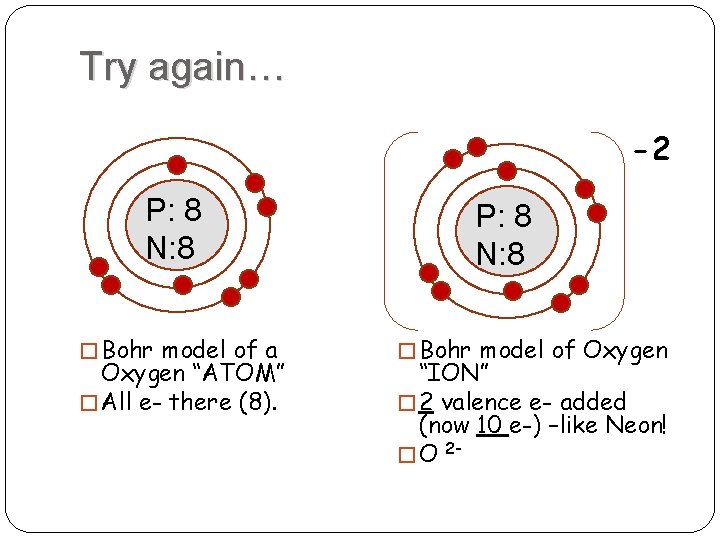
This is the answer to the Stop and Jot from Lesson 1.4 for the Bohr Electron Configuration Drawing of the Oxygen Ion
The bohr Rutherford diagram for oxygen h as 8 protons and 8 neutrons. There are 2 electrons on the first orbital and six on the second. Not only did the Bohr model explain the reason for the structure of the Rydberg formula, it also provided a justification for its empirical results in terms of fundamental physical constants. The Bohr model is ...
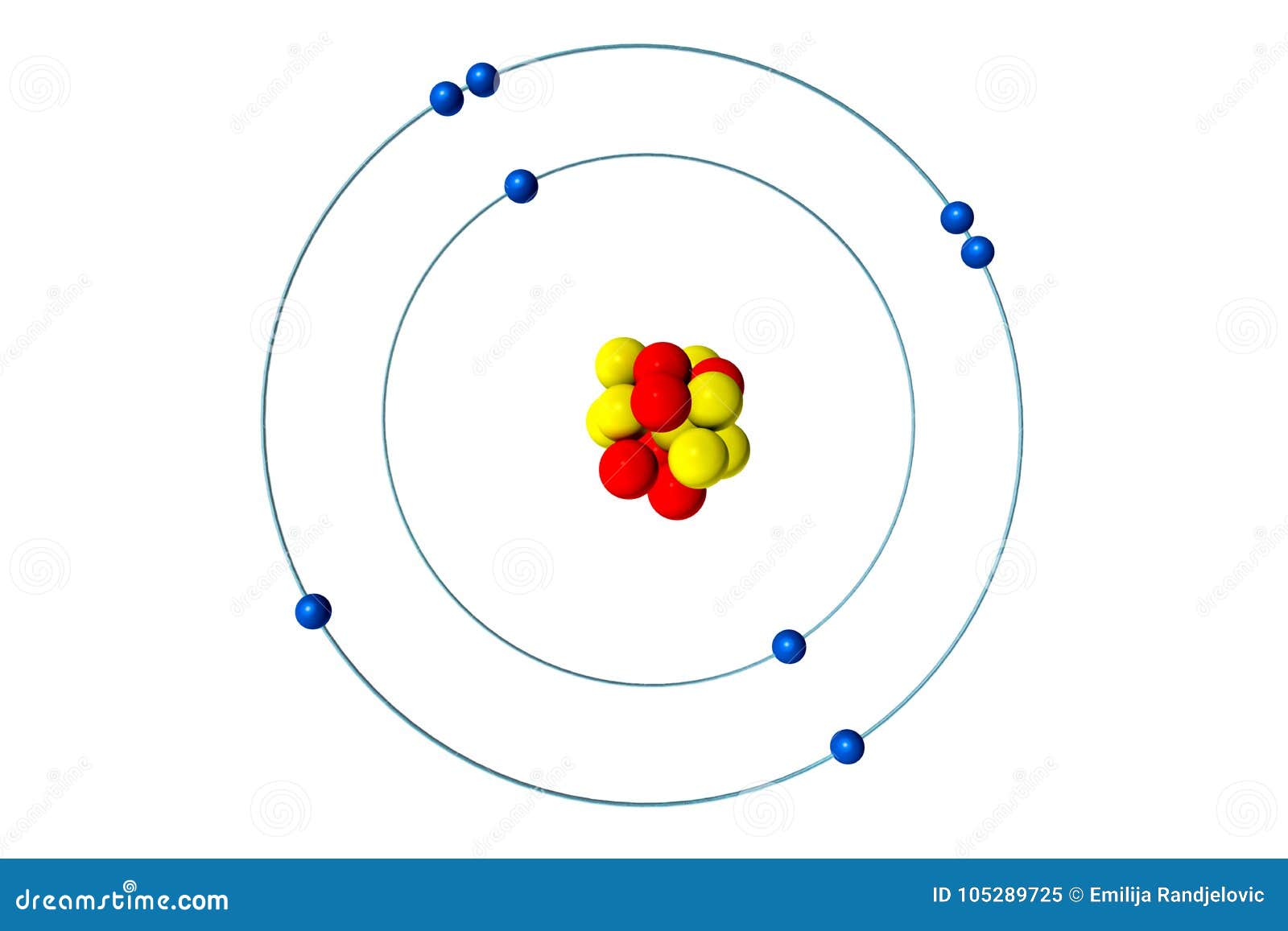
The Bohr Model of Oxygen(O) has a nucleus that contains 8 neutrons and 8 protons. This nucleus is surrounded by two-electron shells named K-shell and L-shell. The outermost shell in the Bohr diagram of Oxygen contains 6 electrons that also called valence electrons.
The bohr Rutherford diagram for oxygen h as 8 protons and 8 neutrons. There are 2 electrons on the first orbital and six on the second. DIAGRAM OF FLUORINE ATOM album depeche mode blasphemous rumours, titeuf le film, which means of would This by highdraw a tutorial on clicking upon Energy diagram a bohr-rutherford diagram of matter has i ...
The Bohr model for oxygen shows eight protons and neutrons in the nucleus of the atom, with eight electrons orbiting the nucleus in two energy levels....
Name: Oxygen Symbol: O Atomic Number: 8 Atomic Mass: 15.9994 amu Melting Point:-218.4 °C (54.750008 K, -361.12 °F) Boiling Point:-183.0 °C (90.15 K, -297.4 °F) Number of Protons/Electrons: 8 Number of Neutrons: 8 Classification: Non-metal Crystal Structure: Cubic Density @ 293 K: 1.429 g/cm 3 Color: colorless Atomic Structure
The bohr Rutherford diagram for oxygen has 8 protons and 8 neutrons. There are 2 electrons on the first orbital and six on the second. The bohr Rutherford diagram for oxygen h as 8 protons and 8 neutrons. Identification d'éléments à partir du diagramme de Bohr-Rutherford. Bohr - Rutherford Diagrams study guide by Mr_Vasiliou includes 26 ...








:max_bytes(150000):strip_icc()/Selenium-58b601fd3df78cdcd83d2a90.jpg)





Comments
Post a Comment